Often, we hear about osmosis in the context of some new technology - water filtration and purification, for instance. The adverts make the process sound like something new and awe-inspiring; a sure confidence booster. It certainly inspires awe, not so much for the process but because of the results. However, it's not some new discovery; our natural world and even our own bodies have been sustaining themselves through osmosis since the dawn of time.
| Osmosis Takeaways | |
|---|---|
| Osmosis and diffusion are similar; | osmosis is distinguished by a permeable barrier |
| Osmosis plays a vital role in biological functions: | water enters and leaves cells by osmosis |
| There are two main factors that affect osmosis: | the concentration gradient and osmotic pressure |
| Osmosis fulfils a primary function: | equalising solute concentrations across a semi-permeable barrier |

What Is Osmosis?
In general terms, osmosis is the act of equalising solute concentrations on both sides of a semi-permeable barrier. If one side has a high concentration of solutes, there must be less water present. If, on the other side of the semi-permeable barrier, there is a lower concentration of solutes - thus a higher potential for water, water molecules will cross the barrier to make the concentration of solutes equal on both sides. 
In biology, osmosis refers to the net movement of water molecules through cell membranes.
We'll focus on osmosis in biology in a bit; for now, let's continue with our general overview of osmosis; namely, the vocabulary specific to this phenomenon.
- osmotic pressure: the minimum pressure needed to prevent inward flow across the semi-permeable membrane
- osmotic gradient: the difference between two solutions' concentration across the membrane
- turgor pressure: the force that presses the plasma membrane within the cell toward the cell wall
- hypotonic: the cell gains water molecules
- isotonic: no net movement of water across the membrane
- hypertonic: the cell loses water molecules
- plasmolyzed: the cell's state when hypertonic
- flaccid: the cell's state when isotonic
- turgid: the cell's state when hypotonic
With this vocabulary laid out, before we move onto the structure of plant and animal cells that permit osmosis, let's look at some factors that may affect the process.
Factors that Affect Osmosis
Two main factors affect the osmotic process: the concentration gradient - also called osmotic gradient, and osmotic pressure. We gave a brief definition of both in the preceding segment; now, let's talk about them in greater detail.
Concentration Gradient
The concentration gradient relates to the concentration of particles dissolved in a solution; particularly, the difference between the percentages of concentration. This condition exists when there is a heavier concentration of solutes on one side of a semi-permeable membrane than on the other. Imagine you've just added a bit of salt to a glass of water. The point where you introduced the salt has a higher concentration gradient than elsewhere in the water and, as it sinks to the bottom and accumulates, that's where the concentration is highest. As you then stir the salt into the water, those molecules disperse, ultimately distributing the salt evenly throughout the water. At that point, the solution is said to have reached equilibrium. Cells use concentration gradients to move a substance in or out of their cells. The stored energy inherent in concentration gradients is used for that purpose. Perhaps the most obvious example of such a usage happens during one of our most vital and fundamental functions: breathing. Granted, it's an instance of gas reflecting a concentration gradient but it can serve well to illustrate the principle. Oxygen follows the rules of concentration gradients, just like solutes do. It too diffuses to areas of low concentration from areas of high concentration. That's how, during their course through our lungs, our blood cells gain oxygen. Later, as those cells course through our bodies, they lose their oxygen content; that gas is again diffusing from a high-concentration area (the cells) to low-concentration areas - our organs and tissues. Find biology tutors here on Superprof. 
Osmotic Pressure
Simply put, osmotic pressure is how hard water has to push to get through a semi-permeable barrier. This pressure is determined by the concentration of solute; water will work harder to access an area with a higher solute concentration. Of course, water has no independent will or knowledge of solute gradients; it merely obeys the laws of nature that dictate the random distribution of matter.
The equation used to calculate osmotic pressure is ? = MRT
That is, indeed, the symbol for Pi but, in this equation, it does not represent that value. It was adopted by concurrence; chemists all agree it serves well to represent osmotic pressure. Solving for ?, in this case, is the same as solving for X in any other algebraic equation. On the other side of the equal sign, we find M (molar concentration), R (ideal gas constant) and T (temperature [kelvin]). You might not have to do any osmotic pressure calculations but you need at least one example that perfectly illustrates the concept. For that, we need to know a little bit about cell biology...

Osmosis in Biology
Have you ever gone on holiday and, upon your return, found your plants all wilted? But then, after giving them water, they've perked up again... that is osmotic pressure at work. Plants use osmotic pressure to keep their shape; the water 'inflates' the cells and give them enough space to function properly. A well-watered plant with vibrant green leaves and plump stems isotonic; one that is withering is hypertonic - the water has moved out of the cells and their concentration of solutes is now too high. When you become dehydrated, the cells in your body undergo the same process: water molecules from muscles and other organs transfer into your blood cells via osmosis because they are hypertonic. That leaves a high concentration of solutes in those cells, a condition that could disturb their function. It would help to know the function of cell structures to fully understand that concept, wouldn't it? Unlike generously watering plants, we cannot simply pour buckets of water down our throats and hope for instant results; the effects could be downright dangerous. Slow rehydration is needed to give our cells time to adapt to osmotic pressure. Find online biology tutor here on Superprof. 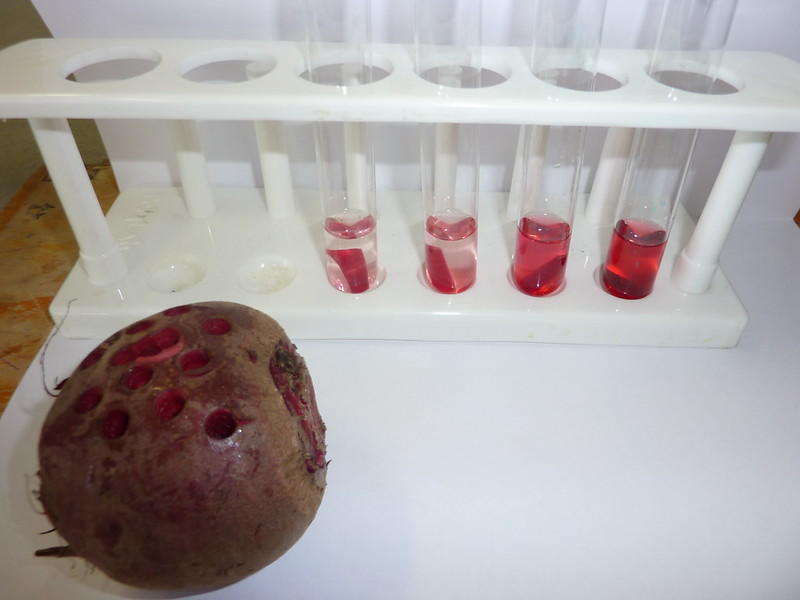
The Potato Osmosis Experiment
Slices (or cylinders) of potato or beetroot in varying degrees of salt or sugar water make for a fun experiment that demonstrates the effects of osmosis. For this experiment, you will need several test tubes - one with just water and the others, with increasingly higher percentages of salt or sugar. The plain-water tube serves as a control; the others demonstrate the speed of water moving out of the potato or beet cylinder. The higher the concentration of salt or sugar, the faster the potato sample will 'deflate'. Soon, you'll see your potato cylinders shrink and, after fishing them out of their test tubes and weighing them, you'll find that their weight has decreased as well. To conduct this experiment, you'll need to cut strips from a potato and dry them off. Set up your test tubes, noting the solute concentration as a percentage on each one. Also, it's a good idea to prepare a table to record your observations. Let your potato or beetroot strips rest in their solutions for 20 minutes and then, pull them out, dry them and take their weight. What were your results? It's not enough to take only one pass at this process; for truly scientific results, you should repeat the experiment to see if you get approximately the same results. Only then have you established proof that the water in the potato cells has moved into the solution to equalise the balance of solutes. Find A level biology courses for adults here on Superprof.
The Difference Between Diffusion and Osmosis
Thumbing through your Biology textbook or referring to Bitesize to review for your GCSE Biology, you might have noticed that 'diffusion' and 'osmosis' see apparently indiscriminate usage. If that's left you scratching your head, you're not alone: many don't quite get the difference between diffusion and osmosis. In principle, they represent the same thing: equalising the concentration of solutes. However, while diffusion may happen under any condition, osmosis takes place only when there is a semi-permeable barrier to cross. Besides the presence of such a membrane, the major difference between diffusion and osmosis is that, in the latter, only water crosses the membrane. In diffusion, both the solute and solvent particles move to equalise the concentration gradient. Keep in mind that, in other fields and other sciences, osmosis takes place using other solutes. However, in biology, the solute in question is only water. In all cases, osmosis may be considered a special type of diffusion, one that requires the presence of a semi-permeable membrane that only the solute may cross. There's lots more to diffusion: its purpose and usefulness in organisms, how it works in plants and animals, and which factors affect diffusion. They're all the subject of another article...
Summarise with AI: